Product Compliance
filing/registration/consulting/training

A cosmetic label is not just a piece of text or a symbol on the outer packaging of a product,It is also the most direct way of communication between consumers and products.Compliance of labels,Directly related to consumers' right to know and safety of use,It also determines the legal operating status of the enterprise in the market.with Regulations on the Supervision and Administration of Cosmetics and Regulations on the Management of Cosmetics Labels Implementation of,Regulatory authorities have increasingly strict requirements for cosmetic labeling,Any labeling that does not meet the standards may result in the product being taken down Punishment and even damage to brand reputation.
In this issue, we will introduce you to the tag section in the registration platform,And how to fill it out.
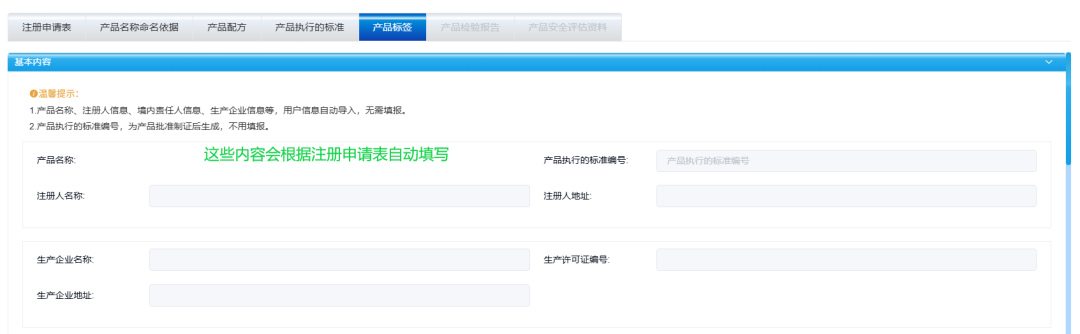
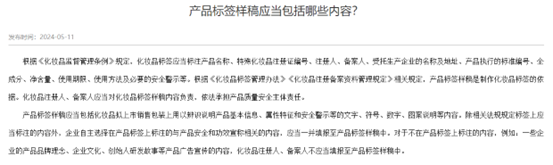
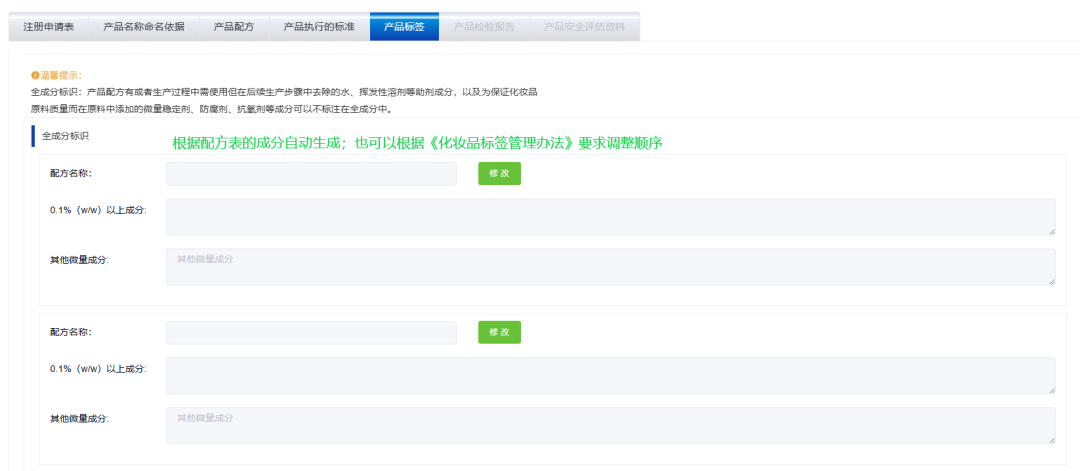
Filling in the ingredients of cosmetics labels
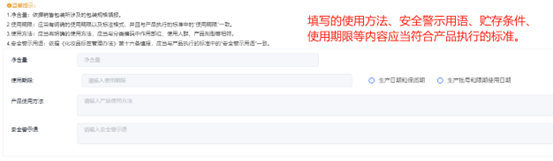
Fill in other content
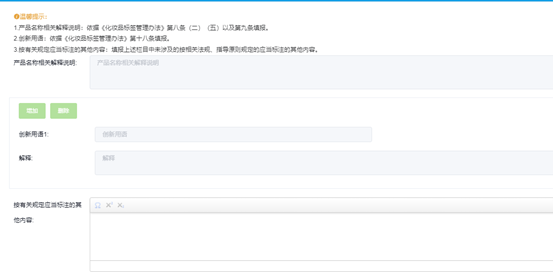
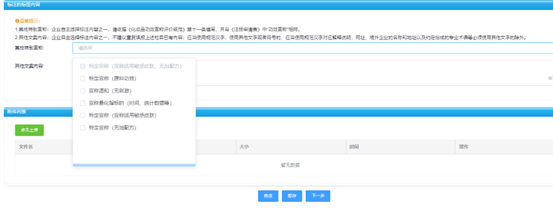
Attention The filling content of the label sample should comply with Regulations on the Management of Cosmetics Labels requirement.
The "Measures for the Administration of Cosmetics Labels" will be officially implemented on May 1, 2022, and will impose more detailed requirements on label content. When preparing sample drafts, companies should focus on the following points:
1.authenticity All label information must be truthful accurate,No false or exaggerated advertising is allowed.for example,Unverified efficacy claims cannot be labeled.
2.integrity The label must contain necessary information,Especially the product name full composition Net Content registrant Production enterprise and other information.
3.normativity The raw material names of the formula ingredients should use the standard Chinese names listed in the catalog of used cosmetic raw materials INCIName or English name:The formula contains new cosmetic ingredients that are still under safety monitoring,Should use registered or filed raw material names.The unit of measurement must comply with national standards,The date identification needs to be clearly stated Production Date or Deadline for use .
4.Risk statement If there are usage restrictions on the product,If it contains hydrogen peroxide Thioglycolic acid, etc,It is necessary to clearly indicate the usage precautions in the label.
5.Disable vocabulary Prohibited The safest option No side effects Instantly effective Absolute language,Avoid misleading consumers.
for example
●Functional ingredients such as nicotinamide hyaluronic acid ,Must be accurately presented in the ingredient list,But it cannot be emphasized solely in promotional language.
If it is imported cosmetics,It is also necessary to indicate the name of the domestic responsible person on the label address.
case example
01

02

03

04

05

frequently asked questions