Product Compliance
filing/registration/consulting/training

When Chinese beauty brands plan to enter the Australian market,The first challenge often encountered is not market competition,But rather a complex regulatory classification problem.Incorrect judgment of product category,Not only will it cause delays in listing,More likely to face product delisting and legal risks.This article will interpret the definition of cosmetics in Australia Main regulatory authorities and relevant regulations.
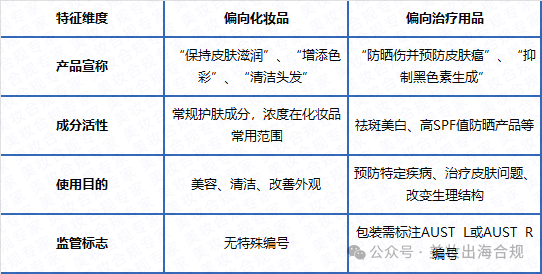
One Core judgment Is your product a cosmetic or a therapeutic product
This is the most critical decision that determines the regulatory path and compliance costs.The legal definition has clear distinctions
According to Australian law,cosmeticspoint
Intended for contact with the outside of the human body,To achieve cleanliness beauty Add fragrance Substances that alter their appearance or protective purpose.
ButTherapeutic suppliesThis includes
Any representation Or it may be understood as Having preventive measures diagnosis Treat diseases or affect body structure/Functional products.
Grey Areas and Key Points for Judgment in Practice
High risk category warning
The following categories are highly likely to be classified as therapeutic products in Australia Sunscreen Products declare Broad spectrum protection or Preventing skin cancer of Anti hair loss products Fluoride containing anti moth toothpaste Promotion ability spot removal and whitening Functional skincare products.
Two regulatory authorities
1. AICIS Industrial Chemicals Import Agency - By evaluating imports or manufacturing introduce Risks associated with industrial chemicals,And promote its safe use methods,To protect the Australian people and the environment.
Under the Australian legal framework,All cosmetic ingredients are considered industrial chemicals.This means
● Any new product formula or ingredient used,We must consider whether it is necessary to passAICISThe evaluation
● Enterprises need to consider the type of introduction Column name introduction Evaluation introduction, etc Complete the corresponding declaration
● Focus on reviewing the health and environmental risks of chemicals
2. ACCC Competition and Consumer Commission - It is a national competitor of Australia consumer Fair Trade and Product Safety Regulatory Agencies.
Responsible for:
● Accurate product labeling Including full labeling of ingredients Warning signs, etc
● The advertising promotion is truthful and not misleading
● The product meets general safety requirements
3. TGA Therapeutic Supplies Management Bureau - Australia is responsible for the evaluation Review and supervise government agencies identified as therapeutic products,Regarding drugs Regulation of medical devices and biological products,To help Australian people maintain health and safety.
This is the strictest regulatory path.When the product crosses cosmetics and Therapeutic supplies When it comes to boundaries,Will faceTGAComprehensive supervision.
Three Corporate compliance recommendations
First: Pre-classification assessment
Compliance assessment should be conducted during the product development phase. Key point:
● Review the product claims in the plan
● Analyze the concentration and function of key components
● Consider hiring a professional compliance consulting firm for pre evaluation
Second: Planning for dual compliance
Even if the target is "ordinary cosmetics", it must:
● Ensure that all ingredients comply withAICISrequirement
● Prepare to meetACCCRequired complete ingredient label
● Avoid any language that may be interpreted as therapeutic claims
Common Misconceptions Warning
✗ Our product is in China Special cosmetics ,But in Australia, it can be declared as regular cosmetics
The classification system between China and Australia is completely different,Need to be evaluated separately
✗ The concentration of ingredients is very low,Will not be used as therapeutic supplies
TGAWe will comprehensively consider and declare Ingredients and product presentation methods
✗ Launch cosmetics first,Once the sales are good, we will make up for itTGAcertification
Once identified as unauthorized therapeutic products,Will face penalties and brand reputation risks
Conclusion
In the highly regulated Australian market,Correct classification and compliance are not only legal requirements,Can better reflect the quality of the brand and the standardization of management behind it.Enterprises should incorporate Australian regulatory requirements into their research and development considerations at the early stage of product planning,Collaborate with professional regulatory consultants,Develop a clear compliance roadmap.
This article is based on the current laws and regulations in Australia,for reference only.next time,We will share the compliance requirements for exporting sunscreen products to the Australian market,Stay tuned