Product Compliance
filing/registration/consulting/training

Receive2022Annual Ethylene Glycol/diethylene glycol EG/DEG The impact of acute kidney injury events caused by pollution,Indonesian Food and Drug Administration BPOM Recently, a series of strict control measures have been introduced.Following the release of the26Regulations require a risk assessment to be conducted,BPOM January 27, 2026 The day has released supporting documents Raw Material List draft.All oral liquid preparations exported to Indonesia High attention should be paid to mouthwash and other related enterprises
01 Background from low risk to strong regulation
According to official statistics from Indonesia,Due to the presence ofEG/DEGAcute kidney injury caused by compound drugs AKI Cases exceeding206rise,The mortality rate is as high as65% .BPOMrealize,Risks not only exist in chemical drugs,It may also exist in natural medicines health supplements In quasi drugs and certain cosmetics .
for this,BPOMYu2025year10The month has officially been issued 2025Year's Day26About Natural Medicines health supplements Regulations on risk assessment of raw material use in quasi drugs and specific cosmetic preparations .
02 Item26Core requirements of Regulation No
natural medicine health products Quasi drug must comply with Indonesian Pharmacopoeia Or international pharmacopoeia standards .
Specific cosmetics must comply with Indonesian Cosmetics Code Or other recognized standards .
2. Mandatory risk assessment Enterprises must conduct a safety assessment of the entire product lifecycle,The evaluation process includes four steps
Hazard identification (Hazard Identification)
Hazard Characteristics Description (Hazard Characterization)
Exposure assessment (Exposure Assessment)
Risk Characteristics Description (Risk Characterization)
03 Focus on Restricted Raw Material List draft for soliciting comments
The following raw materials have been included in the key control list
(Mainly involving the possibility of beingEG/DEG high risk solvents and excipients for pollution)
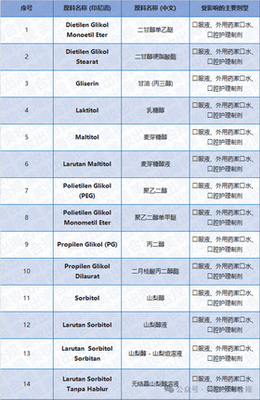
[Data source Draft Attachment Table cite: 223, 224, 226]
The affected product categories are clearly defined as
Natural medicine/health products/Quasi drug Oral liquid Oral External liquid Like mouthwash .
Cosmetics Dental and oral care preparations.
04 Suggestions for enterprise response
2. Upgrade raw material standards Ensure that the above raw materials meet pharmaceutical grade standards Pharmaceutical Grade standard,And can provide corresponding impurity detection reports especially forEG/DEGThe detection .
3. Participate in the solicitation of draft opinions At present, the list of raw materials is still in the draft stage.
Deadline: February 10, 2026
Feedback link https://bit.ly/KP-SKBB
Conclusion